Partners
 University of Lille (Lead partner)
University of Lille (Lead partner) University of Cambridge
University of Cambridge Universiteit Gent
Universiteit Gent Technische Universiteit Delft
Technische Universiteit Delft Lattice Medical
Lattice Medical University College London
University College London ProDigest
ProDigest Universiteit Leiden
Universiteit Leiden Société d’Accélération du Transfert de Technologie, Nord
Société d’Accélération du Transfert de Technologie, Nord Centre Hospitalier Régional Universitaire de Lille
Centre Hospitalier Régional Universitaire de Lille
Observer partners
 Addenbrooke’s Hospital
Addenbrooke’s Hospital Covestro
Covestro Eurasanté
Eurasanté FabRx
FabRx Intract Pharma
Intract Pharma Janssen Pharmaceuticals
Janssen Pharmaceuticals Medtronic
Medtronic Oticon Medical
Oticon Medical Pfizer
Pfizer PKMed
PKMed The Queen Elizabeth The Queen Mother Hospital
The Queen Elizabeth The Queen Mother Hospital Dr. Reddy’s Research & Development B.V.
Dr. Reddy’s Research & Development B.V. Vesale Pharma.
Vesale Pharma.
University of Lille
The research group of Prof. Juergen and Florence Siepmann (http://u1008.univ-lille2.fr) is specialized in the field of advanced drug delivery systems. It is a highly interdisciplinary team with members belonging to the College of Pharmacy, School of Medicine and the University Hospital Lille. Different types of innovative drug delivery systems are prepared, characterized (in vitro and in vivo) and optimized in order to increase the therapeutic effects and decrease the risk of undesired side effects of medical treatments. In the frame of this Interreg project, novel colon targeting systems, inner ear implants and biodegradable microparticles are studied.


Integration, transversality, multidisciplinarity – the Institute for Translational Research in Inflammation (INFINITE U1286, http://lille-inflammation-research.org) headed by Dr. Laurent Dubuquoy, is unique in the world. It conducts an international-level scientific and medical research to understand and to treat inflammatory diseases. INFINITE brings together some 120 researchers organized in open, cross-disciplinary teams to study inflammatory conditions, with the objective to provide new specific treatment solutions for patients. As part of the Site Drug project, we will test the therapeutic efficacy of the novel drug delivery systems in rodent experimental models of Inflammatory Bowel Diseases.
TU Delft
The Biomaterials & Tissue Biomechanics section at TU Delft – Faculty 3mE performs multidisciplinary research on the development of advanced functional biomaterials that replace and/or enable regeneration of damaged tissues. Starting from a clinical need, an advanced design concept or fabrication method, multifunctional structures and surfaces are developed and tested in vitro. Examples include bone implants with regenerative and antibacterial properties (see the figures below), drug eluting stents, scaffolds for cartilage/osteochondral defects. Within the Interreg Site Drug project, the research will explore the 3D printing technology for fabrication of drug eluting inner ear implants. In addition, the mechanical properties of the various implants developed in the project will be assessed based on novel in vitro methodologies and set-ups, relevant for each implant.
 |  |  |  |
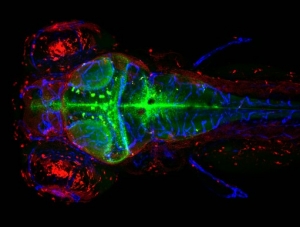
University of Leiden
The Leiden Academic Centre for Drug Research (LACDR) – part of the Faculty of Science – is a centre of excellence for multidisciplinary research on drug discovery and development. Within LACDR, the Biologics Formulation Group chaired by Prof. Jiskoot focuses on the formulation and delivery of biopharmaceuticals, with a special interest in studying the degradation and biodistribution of protein therapeutics and vaccines. For in vivo studies, the group applies the zebrafish embryo model to study the interaction of cells and organs with bio/nanomaterials. Within the Interreg Site Drug project, the research group will develop a zebrafish model for inner ear drug delivery and test the interactions of newly developed (bio)materials with the immune system.
|  |  |
University College London
The UCL research group is specialised in oral drug delivery, digital health, innovative pharmaceutical technologies and translational research. Their research is focused on forging links between basic science and clinical outcomes. They have invented a number of advanced drug products, many of which have been commercialised (Asacol 1600, Yaldigo and Octasa 1600). To date, more than a million patients have benefited from inventions created and developed by the research group. Within the INTERREG project, novel colon targeting systems are being developed using advanced technologies, such as 3D printing.

ProDigest
ProDigest BV is a spin-off company from the University of Gent and was founded in 2008. As a contract research company it has built up a long-term demonstrated expertise in the development and use of unique enabling in vitro models that simulate the complete gastrointestinal tract (GIT). Using its validated SHIME® technology, ProDigest can accelerate and complement the R&D activities and product development of companies in the pharma industry by providing insights into the solubility, the release, the stability and the bioavailability of lead Active Pharmaceutical Ingredients (API) throughout the GIT under highly standardized and biologically relevant GIT conditions. Furthermore, next to adequately simulating the physiological conditions of the GIT, the SHIME® technology platform adequately simulates the ileal and colonic microbiota thereby providing unprecedented insights into possible bi-directional drug-bug interactions. ProDigest is a flexible service provider that is specialized into developing tailor-made experimental designs that can answer the research questions of its customers. As such, ProDigest is continuously optimizing its simulatory models to not only simulate the GIT physiology and microbiota of healthy adults but also that of specific disease states making it possible for pharma companies to perform pre-clinical research with their APIs under GIT physiological and microbiota conditions which are biorelevant for the final targeted patient population. In the frame of this Interreg project, ProDigest will develop an in vitro model allowing to test the efficacy of colon-targeted release systems specifically designed to be administered to patients suffering from inflammatory bowel disease (IBD).

Lattice Medical
LATTICE MEDICAL develops a disruptive bioabsorbable implant for breast reconstruction. We use an original approach combining advantages of actual reconstruction technics and 3D printing to give a simple surgery, more accessible to the patient and less expensive: a bioabsorbable prosthesis. The procedure uses autologous fat tissues injected into an absorbable implant allowing high volume reconstruction in one surgery. In the frame of this intereg project, the integration into the tissue engineering chamber of a compound promoting the growth of the fat flap and reducing the inflammatory risk are studied.
 |  |
SATT Nord
SATT Nord is a Technology Transfer Office designed to work in close collaboration with the academic research laboratories. It is a tool for economic development through innovation. Satt Nord covers the whole value chain of technology transfer: increasing the awareness of researchers to innovation, sourcing high potential inventions in academic research labs, cofunding private-public collaborations, financing proof of concept and prototyping projects, as well as marketing “ready to industrialize” technologies through licensing out agreements, or creating start-up. During the sitedrug project, SATT Nord will make researchers aware of the technology transfer process and will support them in the valorization’s initial stages of the inventions resulting from the project.
 |  |

